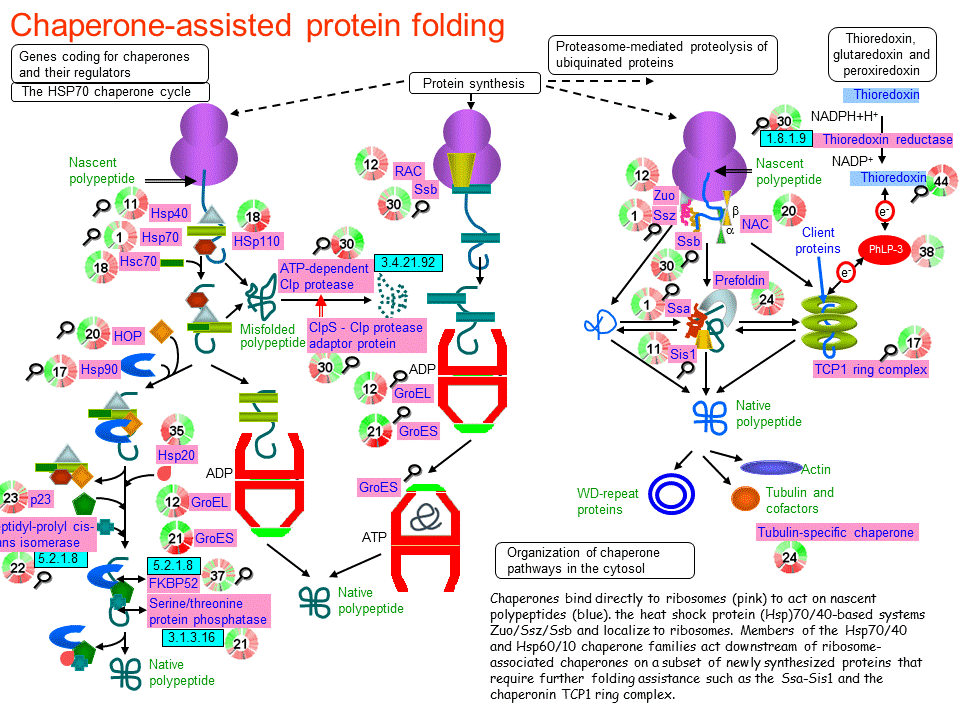 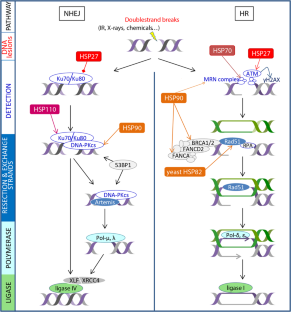
Under normal conditions, HSPs account for 5–10% of the total cellular protein content and they work as an integrated network to maintain proteostasis.Īlthough under extreme conditions, the heat shock transcription factors (HSFs) are activated in response to stresses, leading the transcription of abundant HSPs to buffer the stresses.Īnd based on the primary molecular chaperon function, they also participate in multiple processes in eukaryotic cells, and malfunction of HSPs has reported to related with many diseases. They function in a wide range of cellular housekeeping processes, including the folding of newly synthesized polypeptide, refolding metastable proteins, protein complex assembly, the degradation of misfolded proteins, and dissociating protein aggregates. The ubiquitous and conserved heat shock proteins (HSPs), which exist in both prokaryote and eukaryote organisms, belong to a large family of proteins that are in charge of proteostasis. 
In living organisms, cells are under constantly changing conditions and maintenance of cellular protein homeostasis is crucial for cell survival and integrity,Īs protein misfolding and aggregation have been found to induce malfunction of proteins and lead to various diseases. These research advances offer new prospects of HSPs as potential targets for therapeutic intervention. In this review, we describe the current understandings about the molecular mechanisms of the major HSP families including HSP90/HSP70/HSP60/HSP110 and small HSPs, how the HSPs keep the protein proteostasis and response to stresses, and we also discuss their roles in diseases and the recent exploration of HSP related therapy and diagnosis to modulate diseases. Therefore, malfunction of HSPs is related with many diseases, including cancers, neurodegeneration, and other diseases. In addition to their chaperone functions, they also play important roles in cell signaling transduction, cell cycle, and apoptosis regulation. They function as molecular chaperons in cells and work as an integrated network, participating in the folding of newly synthesized polypeptides, refolding metastable proteins, protein complex assembly, dissociating protein aggregate dissociation, and the degradation of misfolded proteins. HSP protein families are classified based on their molecular weights, mainly including large HSPs, HSP90, HSP70, HSP60, HSP40, and small HSPs. Hsp90 fidelity of translation initiation intrinsically disordered regions molecular chaperone proteostasis.The heat shock proteins (HSPs) are ubiquitous and conserved protein families in both prokaryotic and eukaryotic organisms, and they maintain cellular proteostasis and protect cells from stresses. Our study provides insights into how this abundant molecular chaperone supports a dynamic and healthy native protein landscape. We also discovered that Hsp90 controls the fidelity of ribosome initiation that triggers a heat shock response when disrupted. 
Hsp90 selectively utilized an IDR to regulate client activity as well as maintained IDR-protein health by preventing the transition to stress granules or P-bodies at physiological temperatures. We found that Hsp90 associated with ∼20% of the yeast proteome using its three domains to preferentially target intrinsically disordered regions (IDRs) of client proteins. Using a chemical-biology approach, we characterized the features driving the Hsp90 physical interactome. An essential eukaryotic component of the chaperone system is Hsp90. Molecular chaperones govern proteome health to support cell homeostasis.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
